
Babies made from two women and one man have been approved by the UK's fertility regulator. The historic and controversial move is to prevent children being born with deadly genetic diseases.
The first such child could be born, at the earliest, by the end of 2017.
Some families have lost multiple children to incurable mitochondrial diseases, which can leave people with insufficient energy to keep their heart beating.
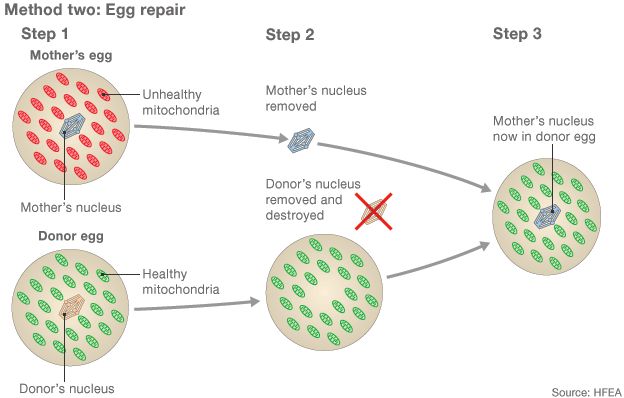
The diseases are passed down from only the mother so a technique using a donor egg as well as the mother's egg and father's sperm has been developed.
The resulting child has a tiny amount of their DNA from the donor, but the procedure is legal and reviews say it is ethical and scientifically ready.
'Historic'
"It is a decision of historic importance," said Sally Cheshire, chairwoman of the Human Fertilisation and Embryology Authority (HFEA)."This is about cautious go ahead, not gung-ho go ahead, and there is a long way to go.
"I'm sure patients will be really pleased by what we've decided today."
But some scientists have questioned the ethics of the technique, saying it could open the door to genetically-modified 'designer' babies.
The HFEA must approve every clinic and every patient before the procedure can take place.
Three-person babies have been allowed only in cases where the risk of a child developing mitochondrial disease is very high.
Clinics can now apply to the HFEA for a licence to conduct three-person IVF.
Donors needed
The team at Newcastle-upon-Tyne Hospitals NHS Foundation Trust and Newcastle University is expected to be the first to be granted a licence.It aims to help 25 couples every year.
Prof Mary Herbert, from the Newcastle Fertility Centre, said: "It is enormously gratifying that our many years of research in this area can finally be applied to help families affected by these devastating diseases.
"Now that that we are moving forward towards clinical treatments, we will also need donors to donate eggs for use in treatment to prevent affected women transmitting disease to their children."
Prof Sir Doug Turnbull, the director of the Wellcome Centre for Mitochondrial Research at Newcastle University, said: "We are delighted by today's decision.
"We will also provide long-term follow up of any children born."
NHS England has agreed to fund the treatment costs of the first trial of three-person IVF for those women who meet the HFEA criteria, as long as they agree to long-term follow up of their children after they are born.
How does it work?
Mitochondrial disease is caused by defective mitochondria - the tiny structures in nearly every cell that convert food into useable energy.One in 4,300 children are born with such severe symptoms they develop muscle weakness, blindness, deafness, seizures, learning disabilities, diabetes, heart and liver failure. It is often fatal.
The aim of the procedure is to get the healthy mitochondria from the donor.
But mitochondria have their own DNA, which is why resulting children have DNA from three people.
However, everything that defines physical and personality traits still comes from parents.
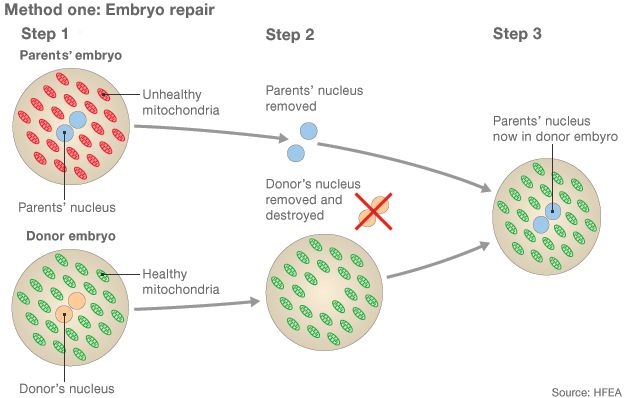
1) Two eggs are fertilized with sperm, creating an embryo from the intended parents and another from the donors 2) The pronuclei, which contain genetic information, are removed from both embryos but only the parents' are kept 3) A healthy embryo is created by adding the parents' pronuclei to the donor embryo, which is finally implanted into the womb
Robert Meadowcroft, from the charity Muscular Dystrophy UK, said: "This historic decision will open the door to the first licensed treatments being offered.
"We know of many women who have faced heartache and tragedy and the sorrow of stillbirths, while trying to start their own family, and this decision gives them new hope and choice for the first time."
Prof Frances Flinter, professor in clinical genetics at Guy's and St Thomas' NHS Foundation Trust, called the decision "wonderful news".
She added: "It is infinitely preferable that the early clinical trials should be done in a tightly regulated system in the UK, with long term follow-up of any children born, rather than in countries where there is no regulation or oversight."
Prof Sir Robert Lechler, president of the Academy of Medical Science, said that the decision means "groundbreaking research can now be translated from theory into practice and transform lives in the clinic".
However, the decision is not universally welcome.
Dr David King, from the campaign group Human Genetics Alert, said: "This decision opens the door to the world of genetically-modified designer babies.
"Already, bioethicists have started to argue that allowing mitochondrial replacement means that there is no logical basis for resisting GM babies, which is exactly how slippery slopes work."
However, the UK will not be the first country in the world to have children born through the three-person technique.
A Jordanian couple and doctors in New York performed the procedure in Mexico and the resulting baby is understood to be healthy.
Don't be afraid to embrace these new technologies when they arrive here. If we can avert tragedies and heartbreak and be sure our babies will be healthy ... it's all good.





What a wonderful idea ... there are so many women that wants a baby that will give it love and keep the baby safe .
ReplyDeleteLove PIC
The technique will remove all genetic diseases that can be handed down through the mother. Most genetic diseases come from the mom. Babies will be born with a clean slate and get off to a healthy start.
ReplyDeleteThanks PIC for putting Maxy's picture on the blog. I never thought of it. He is still the cutest guy.
Good idea
Love PIC